As a pharmacist, it is critical to recognize that infertility is a significant problem. There are a variety of causes and etiologies for female infertility, many of which remain unknown. Almost 13% of women between ages 15-44 seek treatment for infertility each year. There are many infertility medications available today that may be used throughout different stages of the infertility journey, and it should be known that there is no universal protocol.
After and IF a reason for infertility is determined (15% of couples have unexplained infertility), infertility services can be planned from there. The World Health Organization (WHO) designates anovulation into 3 different classes and this determination can help choose a medication for ovulation induction.
Clomiphene is a selective estrogen receptor modulator (SERM) that is FDA-approved for ovulation induction. By exhibiting both estrogen agonist and antagonist effects, it results in increased gonadotropin release through feedback mechanisms. These are peptide hormones, known as LH and FSH, that regulate ovarian function by stimulating the ovaries to produce follicles. Clomiphene is typically dosed for 5 days starting on day 5 of the menstrual cycle. A few adverse effects to watch out for include flushing, vasomotor symptoms, headache, and stomach upset.
Aromatase inhibitors (i.e., letrozole, anastrozole) are another option used for ovulation induction, although not FDA-approved. They were typically used as a second-line option until recent studies have suggested that it may be more beneficial than clomiphene for polycystic ovary syndrome (PCOS) related infertility. Blocking aromatase stimulates gonadotropin release through feedback mechanisms from reducing serum concentrations of estradiol. Aromatase inhibitors bring a few more side effects to the table, including edema, sweating, GI upset, arthralgias, and fatigue. However, the risk of adverse events is reduced as it is typically only dosed for 5 days during a menstrual cycle.
Recombinant gonadotropin (LH/FSH) injectable products may additionally be used for ovulation induction, however therapy requires close hormonal (i.e., serum estradiol) and sonographic monitoring to ensure follicle production. Gonal-F (FSH) and Follistim (FSH) are commonly used products. Menopur (FSH and LH) is also available for patients who have low LH levels. Patients are typically instructed to inject gonadotropin therapy a few days into their menstrual cycle and to follow up with their clinic routinely for dose adjustments if needed based on serum estradiol levels and ultrasound follicle measurements. Once a follicle appears to be mature, they may be instructed to inject human chorionic gonadotropin (hCG) on days 7, 10, and 13 following ovulation, which acts similarly to LH as an ovulatory trigger. A few products available include Pregnyl, Novarel, Ovidrel, or generic hCG. Sometimes ovulation induction is all that is needed, but oftentimes couples will go on to pursue an intrauterine insemination (IUI) or a complete assisted reproductive technologies (ART) regimen.
A concern to be aware of when using infertility medications is the risk of multiple births, which is <10% for those who use oral agents (i.e., clomiphene, letrozole) and up to 36% for those using gonadotropin therapy. Another serious concern is the risk for ovarian hyperstimulation syndrome (OHSS), which is 1-5% of cycles for those using gonadotropin therapy. The more severe cases involve ovarian enlargement, ascites, electrolyte imbalance, and hypercoagulability that is potentially life-threatening. These complications remind us to use gonadotropins at the lowest dose and duration necessary.
Again, infertility protocols will vary based on who you ask and where you are in the world. In countries outside of the U.S., pulsatile administration of gonadotropin-releasing hormone (GnRH) is indicated for some types of infertility and is given via infusion pump. GnRH triggers the production of FSH and LH. In the U.S., injectable GnRH agonists (i.e., Lupron) and GnRH antagonists (i.e., Ganirelix, Cetrotide) are commonly used as adjuvant therapy. The goal is to allow for lower doses of overall gonadotropin therapy and prevent an LH surge (with subsequent early release of an egg). However, the data to draw conclusions on this as adjuvant therapy is limited. Estrogen and progesterone therapies are frequently added in more complex ART regimens to mimic the hormones of maintaining a pregnancy. Prior to any of the medications mentioned above, you may also see metformin used in PCOS patients or bromocriptine used in hyperprolactinemic patients for ovulation induction.
The graphs below provide a general understanding of the hormone patterns required to induce and maintain a pregnancy, which helps provide a reason for the medications used.
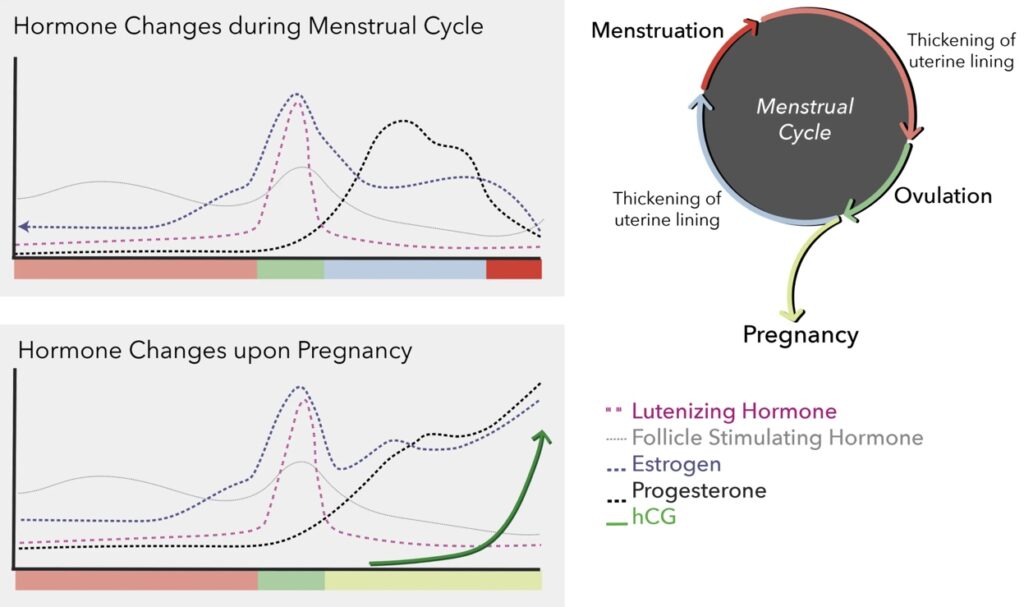
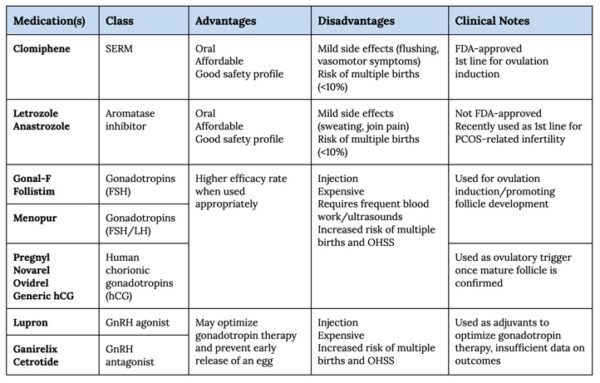
Infertility journeys can be long and difficult. It is important for pharmacists to encourage individualized medication therapy to promote the best outcomes. Please refer to the table below for some advantages and disadvantages of the most commonly used infertility medications.
- 30 medication mistakes PDF
- 18+ Page Drug Interaction PDF
- 10 Commandments of Polypharmacy Webinar based on my experiences in clinical practice
This article was written by Hannah Wetter, PharmD Candidate in collaboration with Eric Christianson, PharmD, BCPS, BCGP
Resources:
Fauser, B., et al. Overview of Ovulation Induction. UpToDate [Web]. Wolters Kluwer Health. Waltham, MA. Last updated November 2021. Accessed via https://www.uptodate.com/contents/overview-of-ovulation-induction?sectionName=GONADOTROPIN%20THERAPY&search=infertility&topicRef=5448&anchor=H14&source=see_link#H523302280
Fauser, B., et al. Patient education: Infertility treatment with gonadotropins (Beyond the Basics). UpToDate [Web]. Wolters Kluwer Health. Waltham, MA. Last updated April 2021. Accessed via https://www.uptodate.com/contents/infertility-treatment-with-gonadotropins-beyond-the-basics
Gonadotropins. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Last updated March 26, 2018. Accessed via https://www.ncbi.nlm.nih.gov/books/NBK548856/
Reproductive Health; Infertility. Centers for Disease Control and Prevention. Last reviewed April 13, 2021. Accessed via https://www.cdc.gov/reproductivehealth/infertility/index.htm
Tyssowski, K. Pee is for Pregnant: The history and science of urine-based pregnancy tests. Harvard University. Published August 31, 2018. Accessed via https://sitn.hms.harvard.edu/flash/2018/pee-pregnant-history-science-urine-based-pregnancy-tests/



This is so helpful! Thank you!
Another very useful drug for treatment of infertility is Carbegoline. It is dopamine receptor agonist that is particularly useful for cases of hyperprolactinemia-induced infertility.
I like it because there are lesser side effects with its use compared to bromocriptine.