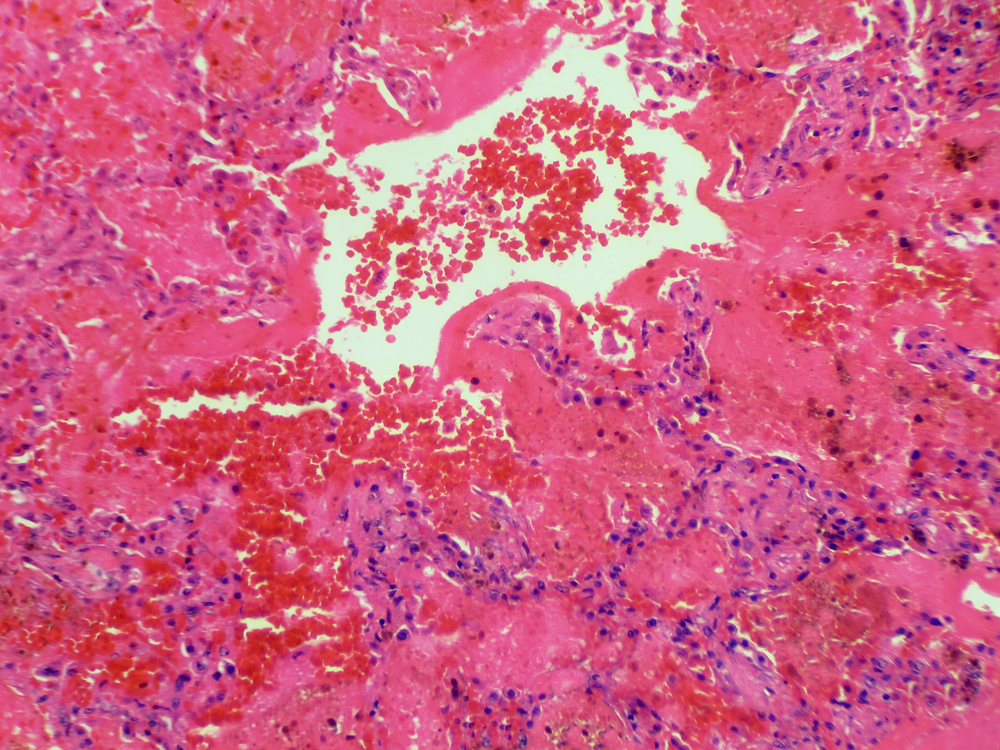
A 21 year old African American male patient with no pertinent medical history presented to the ED by EMS in pulseless electrical activity (PEA) cardiac arrest. As per family at bedside, patient had been having cough with no sputum, headache, fatigue, bloody diarrhea, some chest pain, shortness of breath, and signs of dehydration for the past 3-4 weeks. On the morning of presentation to ED, patient’s sister noticed that he had fallen on his way to the bathroom and began foaming at the mouth. After EMS responded, patient lost pulses en route to the ED. CPR was initiated, 3 rounds of epinephrine were given, and intubation occurred upon ED arrival. Urine toxicology screen was positive for marijuana and benzodiazepines. Patient was found to be in respiratory failure and bronchoscopy showed blood clots in bronchioles. Moreover, patient was in renal failure, resulting in a serological work-up to determine the cause.
Patient was given a total of 6 plasmapheresis sessions, and then continued on intermittent hemodialysis. The medical team following the patient, as well as renal and rheumatology consulting physicians had high suspicions for Goodpasture’s syndrome (GS). Anti-glomerular basement membrane (anti-GBM) antibodies were drawn, with the first two yielding weakly positive results.
GS is a rare organ-specific autoimmune disease that is mediated by anti-GBM antibodies. It typically presents as acute renal failure caused by a rapidly progressive glomerulonephritis, accompanied by pulmonary hemorrhage. The incidence of GS is estimated to be 1 case per million per year, but it is a cause of acute renal failure in approximately 20% of all cases of rapidly progressive or crescentic glomerulonephritis. The exact cause has not been able to be determined, however, certain respiratory infections may trigger the disease and exposure to hydrocarbon fumes, metallic dust, tobacco smoke, or substances such as cocaine may increase risk. Furthermore, symptoms could start out slowly, gradually affecting the lungs and the kidneys, or may become rapidly severe in a matter of days. Initial symptoms may include fatigue, weakness or lethargy, nausea and/or vomiting, loss of appetite, and pale appearance [1-2].
Diagnosis of GS is made through the detection of circulating anti-GBM antibodies; however, kidney biopsy provides definitive diagnosis. GBM antibodies detected by immunoassay have been reported to be highly specific for GS. The sensitivity of this test approaches 87% in untreated patients with systemic disease. A positive test for anti-GBM cannot be relied upon exclusively to establish the diagnosis. Weakly positive test results (5-30 EU/mL) may occur in other immune-mediated diseases, in patients who do not have anti-GBM antibody-mediated disease [3]. In this patient, of the four anti-GBM tests done, their highest positive titer was 2.1 (>1.0 AI: Antibody Detected), representing a weakly positive result.
As for treatment, the 3 principles of therapy in anti-GBM disease are to rapidly remove circulating antibody by plasmapheresis, to stop further production of antibodies using immunosuppression with medications, and to remove offending agents that may have initiated the antibody production. Standard therapy consists of the combination of high doses of glucocorticoids, cyclophosphamide and plasma-exchange, which are all what this patient received [1-2].
Ultimately, after a lengthy hospital stay, this patient’s condition improved. However, they were still reliant on hemodialysis. A renal biopsy was necessary, but patient kept rescheduling and refusing, therefore the patient was discharged with an appointment for follow-up in the rheumatology clinic. So was this a presentation of Goodpasture’s syndrome? Although their anti-GBM antibody was positive, titers were very low. GS is technically possible, but probably unlikely. References
Submitted By: Kayla Torppey, PharmD-PGY 1 Pharmacy Practice Resident in collaboration with Donna M. Lisi, PharmD, BCPS, BCPP, Barnabas Health Care System, South Plainfield, NJ


MED ED 101 IS DOING AVERY GOOD SERVICE IN THE PROCESS OF CONTINUING MEDICAL EDUCATION.
Great contributors have made this website what it is today! Thanks for the comment 🙂
Nice mix of reviews and more complicated cases! I’ll be recommending all my interns search you out, for sure!
Thanks Emma, it’s because of folks like you that this page has grown beyond my wildest expectations!
Yeh looks like it see a lot of wegener polyangyitis here and the ddx usually include GPS initially