by Eric Christianson | Apr 12, 2023 | Endocrine Medication and Disease State Clinical Pearls
It is theorized that patients who are taking a GLP-1 agonist are at a greater risk for pancreatitis due to the medication causing an increase in stimulation of GLP-1 receptors located in the pancreas. This could lead to an overgrowth or overactivation of pancreatic...

by Eric Christianson | Jan 8, 2023 | Clinical Guideline Updates For Pharmacists
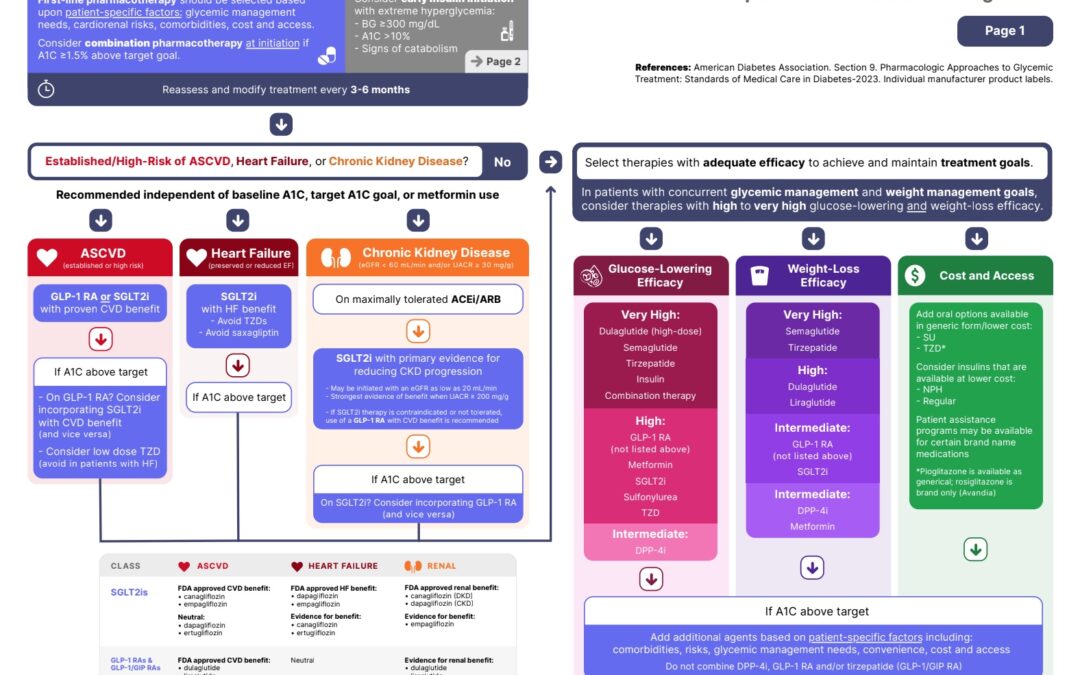
The ADA has released its 2023 guidelines. There are a significant number of impacts on medication therapy, and in this post I will outline my most important takeaways. I was also recently catching up with my friend and fellow pharmacist, Derek Borkowski who is the...

by Eric Christianson | Sep 7, 2022 | Endocrine Medication and Disease State Clinical Pearls
Drug interactions don’t come to the top of my mind when I see GLP-1 agonists in use. We are fortunate that they do not have a lot of significant interactions, but I don’t think you should forget about them altogether. I’ve provided a list below of my...

by Eric Christianson | May 25, 2022 | Clinical Pharmacy News and New Drug Updates, Endocrine Medication and Disease State Clinical Pearls, Medication and Disease State Clinical Pearls For Pharmacists
On May 13, 2022, the U.S. Food and Drug Administration (FDA) approved Eli Lilly and Company’s Mounjaro (tirzepatide), a once-weekly injection for the treatment of type 2 diabetes mellitus (T2DM). Pathophysiology of Type 2 Diabetes Mellitus Incretins are the...

by Eric Christianson | Jan 12, 2022 | Clinical Guideline Updates For Pharmacists
The American Diabetes Association released their 2022 Diabetes Guidelines update. While I wouldn’t call the changes earth-shattering, there are some items in relation to pharmacotherapy that I feel are really important to note. The patient population that I work...